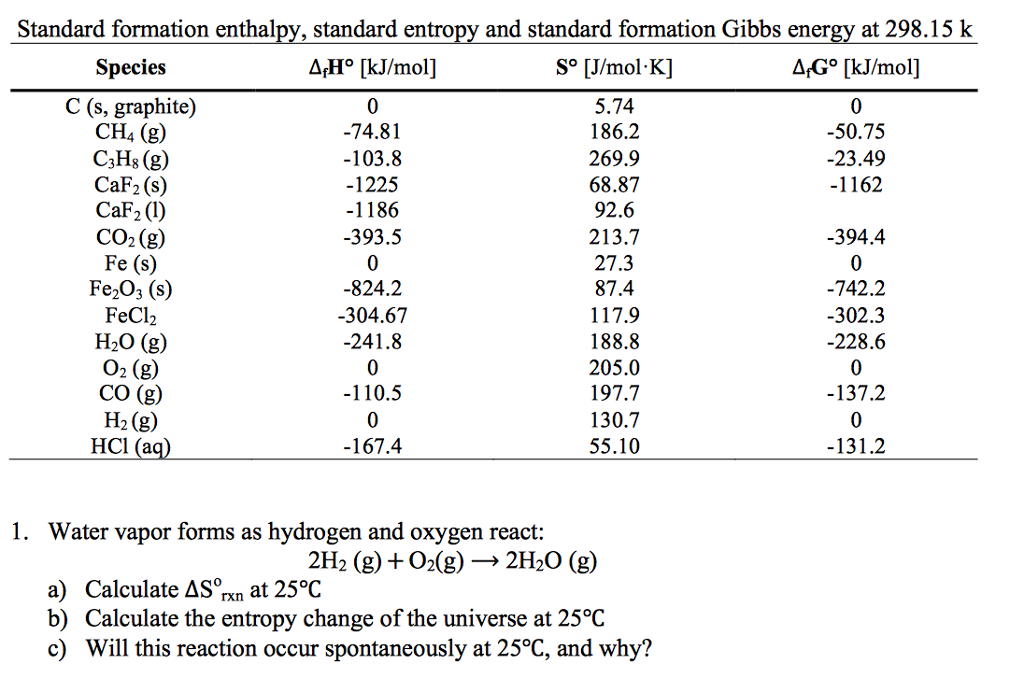
The greater the mass of the molecule, the greater will be corresponding constituents. #= "19.5 cal/mol"cdot"K"cdotln("457.15 K"/"386.75 K") xx "4.184 J"/"cal"#Īnd of course, this is the same idea as before. Molar entropy also depends on the molecular mass of the molecule. Here we assumed that the heat capacity was a constant in the temperature range, "C" -> "C") ~~ barC_P int_("386.75 K")^("457.15 K") 1/TdT# This would give #"14.434 J/mol"cdot"K"#.Īt any phase equilibrium, #DeltaG = 0# so that #DeltabarH_"fus" = TDeltabarS_"fus"#. and DS are both positive and the liquid or gas has more entropy than the solid or liquid. this #barC_P# expression is only good up to "C"#, and then it underestimates #barC_P# by up to #"9 J/mol"cdot"K"# at "C" -> "C") = "4.184 J"/"cal"int_("298.15 K")^("386.75 K")13.07/T + 3.21 xx 10^(-4)(1 - 25/T + 273.15/T)dT# The entropies of gases are much larger than those of liquids. The heat absorbed when one mole of a substance melts at its melting point reversibly is the molar heat of fusion. In order to determine the substance that is expected to have the higher standard molar entropy (S°) at 25 ☌ based from the given pair of substances, analyze the explanation below. [By the way, I compared with a proper heat capacity curve. You'll have to verify whether that is true or not. 
I assume the equation for #barC_P# is in terms of "C"# and not #"K"#. Std entropy change of fusion, fus S o +31 J/(mol·K) Std enthalpy change of vaporization, vap H o +42.3 ± 0.4 kJ/mol: Std entropy change of vaporization, vap S o: 109. HEATING THE "C" -> "C") = "C") barC_P/TdT# Entropy of gas at standard conditions (1 bar) Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific. One way of calculating S for a reaction is to use tabulated values of the standard molar entropy (S°) The entropy of 1 mol of a substance at a standard temperature of 298 K., which is the entropy of 1 mol of a substance at a standard temperature of 298 K the units of S° are J/(mol·K). We break this into the four steps shown above. Calculating S from Standard Molar Entropy Values. #((delbarS)/(delT))_P = (barC_P)/T = 1/T((delbarH)/(delT))_P# This equation, applied to molar volume, molar internal energy and molar entropy, is as follows: VE 5 V 2 VID (9.11) UE 5 U 2 UID (9.10) SE 5 S 2 SID (9.19). Therefore, and you should already know this definition, #dS(T,P) = ((delS)/(delT))_PdT + cancel(((delS)/(delP))_TdP)^(0)#Īnd we know that #((delbarH)/(delT))_P = barC_P#, where #barY# indicates a molar quantity. All of them have tetrahedron structure and the four corners are identical so there is only one way to organize CHF3. 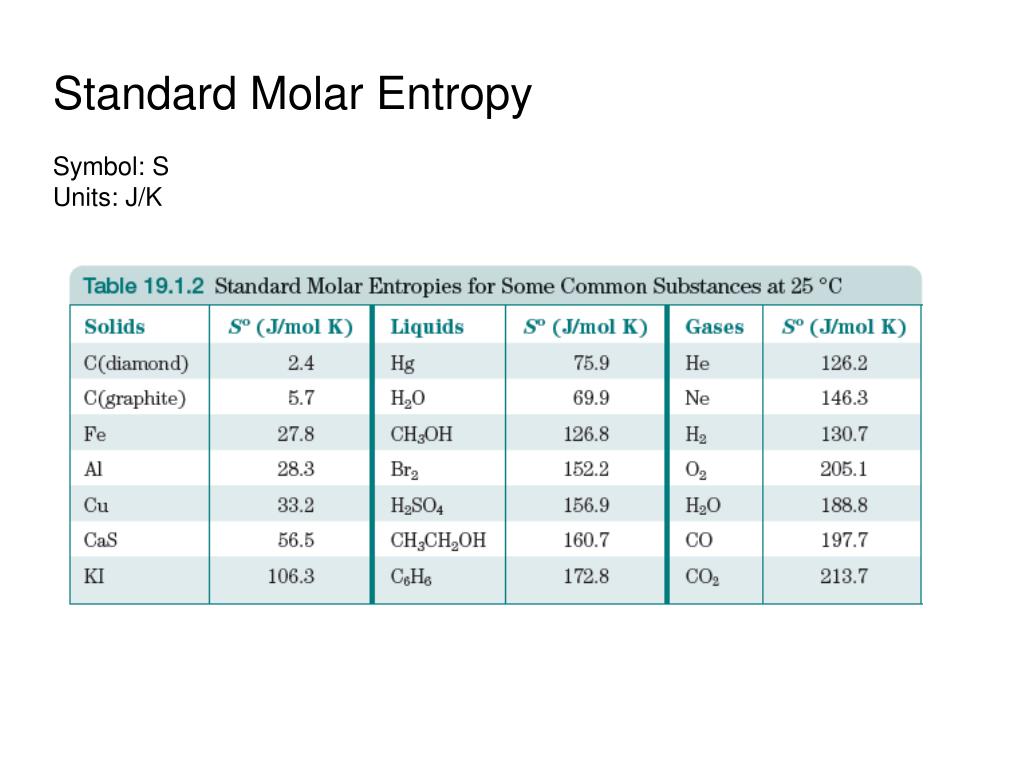
And that makes sense, that the entropy term is going to make our Gibbs free energy more positive. The trend here is based on molecular complexity. And so now our Gibbs free energy right here is going to be minus 890 kilojoules minus 290- so the minus and the minus, you get a plus. White Rose Research Online URL for this paper. Assume the change is reversible and the temperature remains constant.I'm assuming you mean to go all the way through the vapor phase. Re: Arrange in order of increasing standard molar entropy. This is a repository copy of Estimation of standard molar entropy of cement hydrates and clinker minerals. 
Determine the change in entropy (in J/K) of water when 425 kJ of heat is applied to it at 50 oC.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
